Join the Movement to Protect Patients.
Rx-360 offers superior value, support, and services to our members to ensure patient safety. As a nonprofit, Rx-360 contributes to member organizations’ corporate social responsibility efforts, aiding them to operate in an economically and socially sustainable manner.
How We’re Improving the Supply Chain of Pharmaceutical Products
Our Auditing Program and Licensed Audit Reports
Members have access to Rx-360’s Joint Audit Program, which minimizes on-site audits for suppliers, streamlines audit function efficiency, and improves audit quality and thoroughness using industry resources. All ongoing audits are viewable in real-time and all audit reports generated through the Rx-360 Audit Program can be utilized by licensees.
How We Identify Supply Chain Issues in the Pharmaceutical Industry
Supply chains have become more global, complex and increasingly vulnerable to adulteration and counterfeiting. To combat this, Rx-360 has increased its focus on strong partnerships between pharmaceutical manufacturers and suppliers to ensure the integrity of supply chains through on-site auditing. The audit process is completely transparent to members, showcasing supplier and sponsor review and critical audit findings to flag any potential issues.
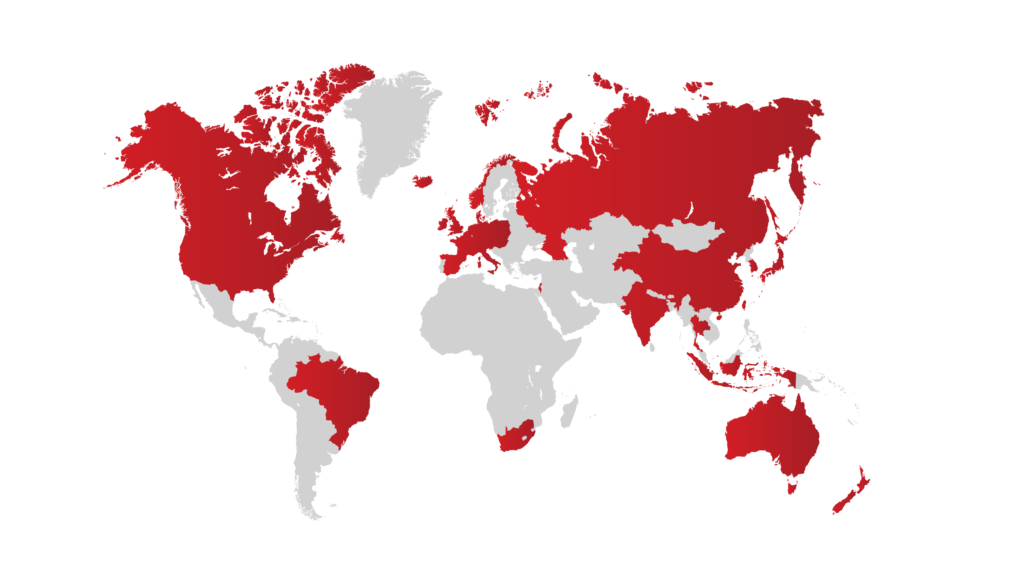
Since Our inception, we’ve worked across the globe
How We Deliver Greater Pharma Patient Safety
Rx-360 prioritizes patient safety by gathering industry peers to share ideas in a confidential environment, improve knowledge base and preparation within the industry, drive industry-wise standards and guidelines, identify and mitigate global supply chain risks, and improve audit quality and thoroughness. Join the movement today.
CONNECT AND COLLABORATE
WITH THE BEST PHARMACEUTICAL
THOUGHT LEADERS ON EARTH
- Ensure there are no bad actors in your supply chain.
- Gain best practices from top pharmaceutical companies.
- Band together to protect patient safety.

One consortium dedicated to
ensuring supply chain integrity
and protecting patient safety
BECOME A KEY PLAYER IN PROTECTING PATIENT SAFETY
"*" indicates required fields






















