Improve Supply Chain Security: An Overview of Our Supplier-Led Working Group’s Whitepapers
In the pharmaceutical industry, ensuring the integrity of the supply chain, material quality, and patient safety is paramount. One of the tools that organizations use to achieve these goals is the audit process. Knowing the […]
Rx-360 Releases Supplier Assessment Questionnaire Version 3.0 as a Resource to the Pharmaceutical Industry
Rx-360 is pleased to announce that the consortium membership led by key members of the Supplier Quality Working Group has released Version 3.0 of the Supplier Assessment Questionnaire.
Rx-360 Collaborates with Michigan State University to Offer Course on Pharmaceutical Brand Protection and Supply Chain Integrity
Rx-360 is pleased to announce that the consortium is collaborating with Michigan State University’s Eli Broad College of Business to develop and offer a new educational course on pharmaceutical brand protection and supply chain integrity.
Rx-360 Expands Trademark Portfolio to Japan
Rx-360 is pleased to announce that the consortium has now expanded their trademark registration for Joint Audit Program® logo to now include Japan.
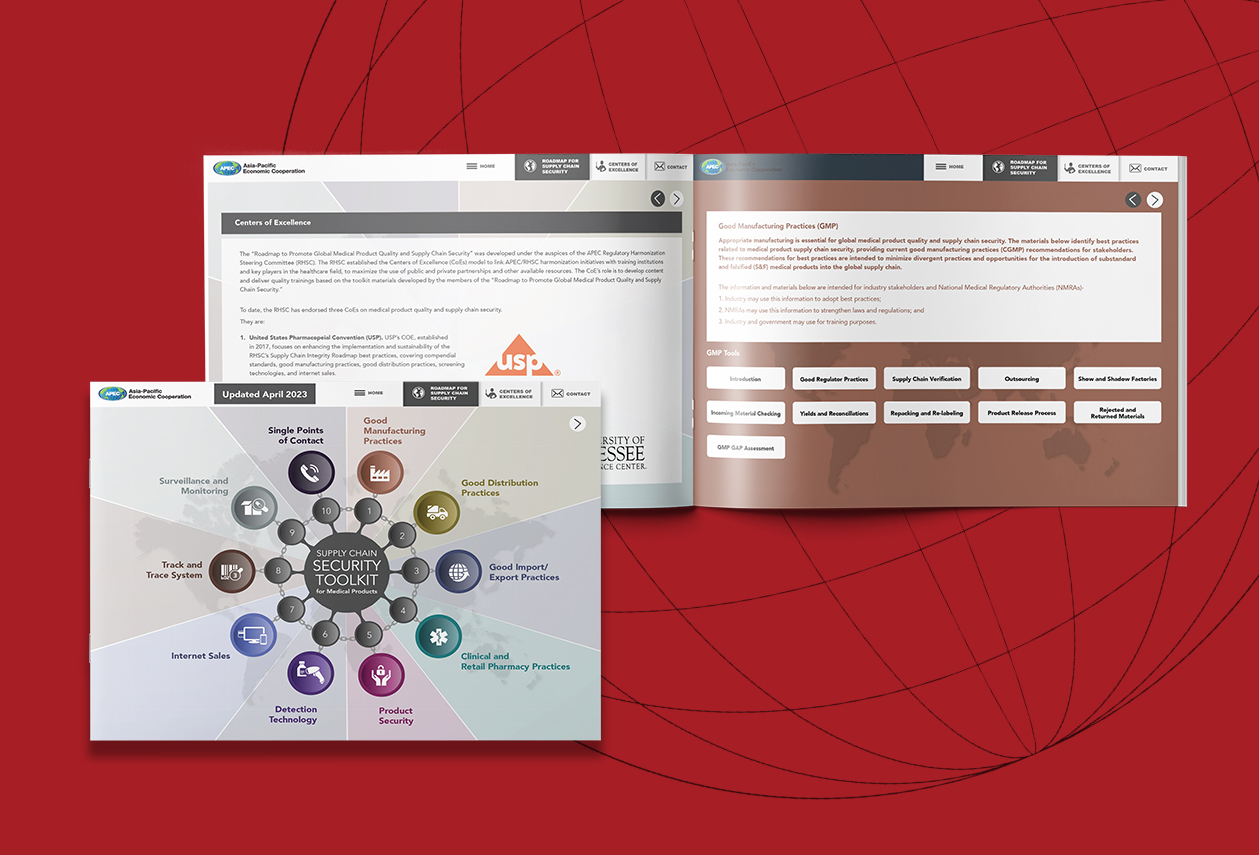
APEC Supply Chain Security Toolkit for Medical Products
Rx-360 has been a promoter of the APEC Supply Chain Toolkit from its founding, which our members played an integral role in. The APEC Supply Chain Security Toolkit is now hosted by the USP. USP […]
Rx-360 Helps NewAge Industries Reduce Audit Costs and Save Time
NewAge Industries, a member of Rx-360, is a leading independent, employee-owned manufacturer of fluid transfer systems that produces high-quality tubing, hose, fittings, clamps, and accessories and offers expert project guidance and services. NewAge Industries […]
Good Distribution & Warehouse Practices for Warehouse & Transportation Suppliers in LATAM
The warehousing and distribution of regulated medical products is a very important part of the supply chain from manufacturer to the patient. Various separate entities are responsible for the storage and shipping of these products […]
Episode 6: Interview with Ann Jordan, CEO of ASQ
Host Jim Fries speaks to Ann Jordan, CEO of ASQ in the sixth episode of The Patient Safety Podcast about recent changes in quality management since the COVID-19 pandemic. Ann discusses how these quality changes […]